Managing the data jungle
Many biology labs fight with a glut of measurement data. New software aims to make this a thing of the past: it simplifies laboratory experiment evaluation and unifies how data is saved. It even identifies measurement errors on the spot.
During laboratory testing, countless measurement results accrue. To completely and systematically archive this body of data is extremely time consuming. In fact, researchers in the life sciences spend a quarter of their time managing data, according to an online survey of 70 people working in biology laboratories conducted by the Fraunhofer Institute for Applied Information Technology FIT in Sankt Augustin. Many of those surveyed reported that they have no centralized or structured approach to data collection in their workplace. And when a PhD student or assistant with years of experience leaves the institute and the successor tries to find and make sense of previous results, the search often begins with cryptic Excel tables and stacks of paper.
The FIT has taken steps to alleviate this problem. With its step-by-step operation, its “MPlexAnalyzer” software makes it considerably easier to manage data. Initially, the FIT experts concentrated on cytometric devices, which enable the simultaneous determination of a variety of proteins in a test batch. This approach, also called a cytometric multiplex assay, is a standard method in every biology lab. However, assays such as these are very complex and produce huge amounts of data, so it is no wonder that personnel without access to any supporting software often lose the upper hand with data collection. The FIT software’s wizard assistant guides users through the entire measurement process, starting with the selection of the microtiter plates, the choice of samples, and the assignment of standard samples, and provides a clearly arranged PDF report for print out; the process is transparent and easy for beginners to grasp quickly.
Antibody coated beads
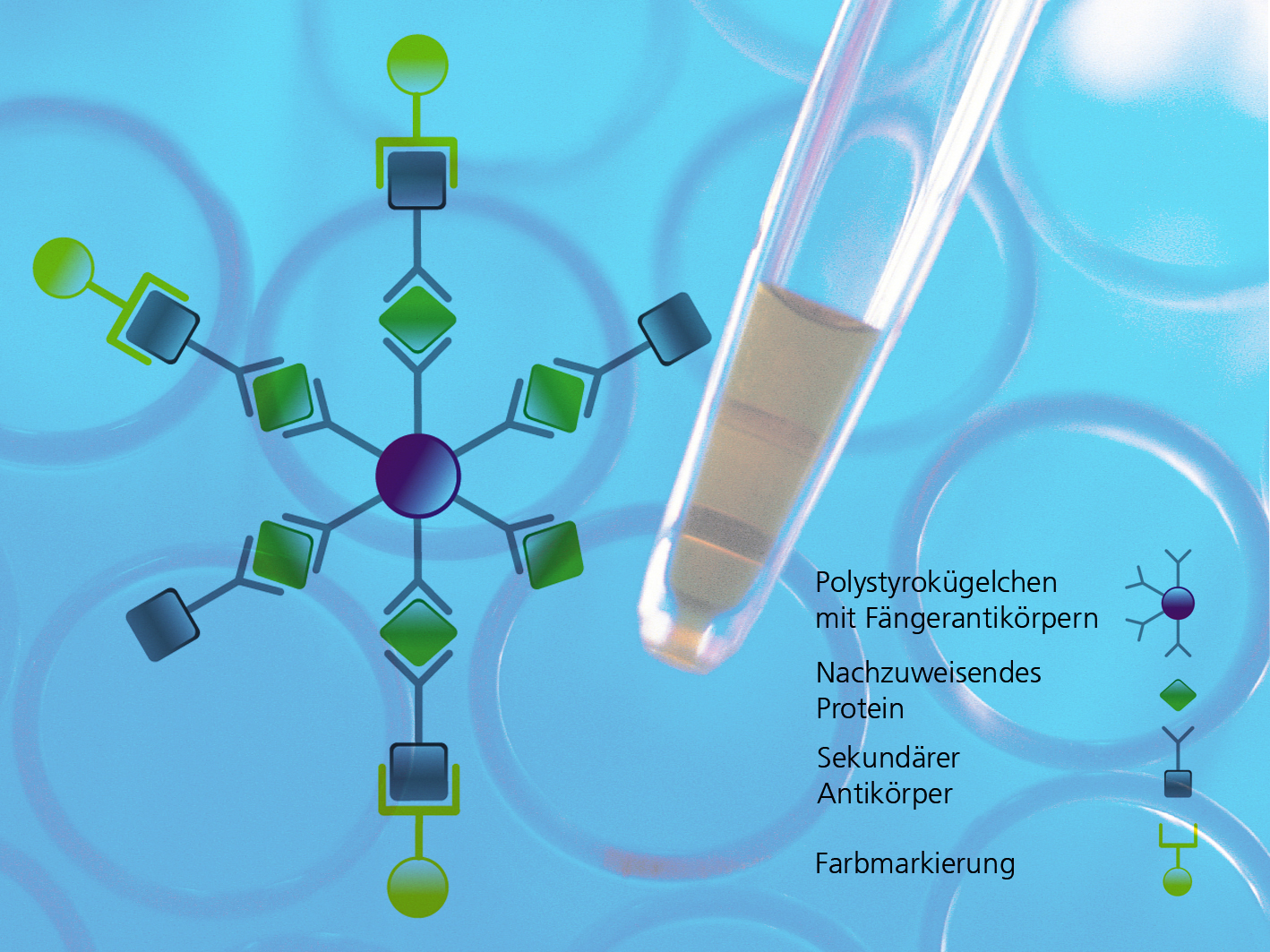
A cytometric multiplex assay uses polystyrene beads with a diameter of only six micrometers. These are infused with a dye mixture that glows when subjected to two laser beams. The glowing pattern is like a fingerprint; up to a hundred different types of beads can be differentiated in this way. During the measurement, the beads flow through a thin glass cannula like pearls strung on a necklace. Cameras measure the color patterns, counting and sorting them as they go past. But biologists are primarily interested in the beads’ second cargo: color-coded antibodies on the surface of each bead that are stimulated by one of the two lasers. This dye emits light at another wavelength – but only if specific substances have bound with the antibodies, such as blood components, cell excretions or cancer cell signal proteins. A multiplex assay examines up to 100 of these substances simultaneously.
The result is a huge data set that records the number of registered beads together with the identified substance. Measurements are highly automated – every minute, up to 96 different samples are tested, each in a little well on a glass plate. One portion of each plate is covered with samples, and another portion with reference substances used to calibrate the readings. Until now, the process of documenting where each sample is located and which measurement is recorded was a time-consuming manual task. “Our software wizard simplifies the process. With just a few mouse clicks, you can mark on the screen which wells contain reference substances or which ones are empty. If the corresponding field is red, then the specification and the measurement don’t match. This means the lab assistant will see immediately if a mistake has been made or if the quality of the measurement is insufficient to reach a statistically reliable conclusion,” explains Dr. Andreas Pippow, a scientist at the FIT.
For the FIT, the software is a way in to the data management market for biological labs. The idea will now be carried over to other applications, such as microscopes. At the biology lab of Fraunhofer FIT’s Life Science Informatics department in Sankt Augustin, scientists are building special microscopes that can automatically shuttle large samples back and forth under the objective and scan them. There are plans for a database of all lab-performed measurements, such as those from multiplex assays, microscope images or other measuring equipment. The appeal of this common data management approach lies in the built-in ability to perform cross checks. For instance, when body cells emit certain chemical messengers as a result of a disease, often these will have consequences for the tissue structure. But the only way to spot this is by using the software to match the signal substance tested in the multiplex assay against microscope images.