Health Research in 4D


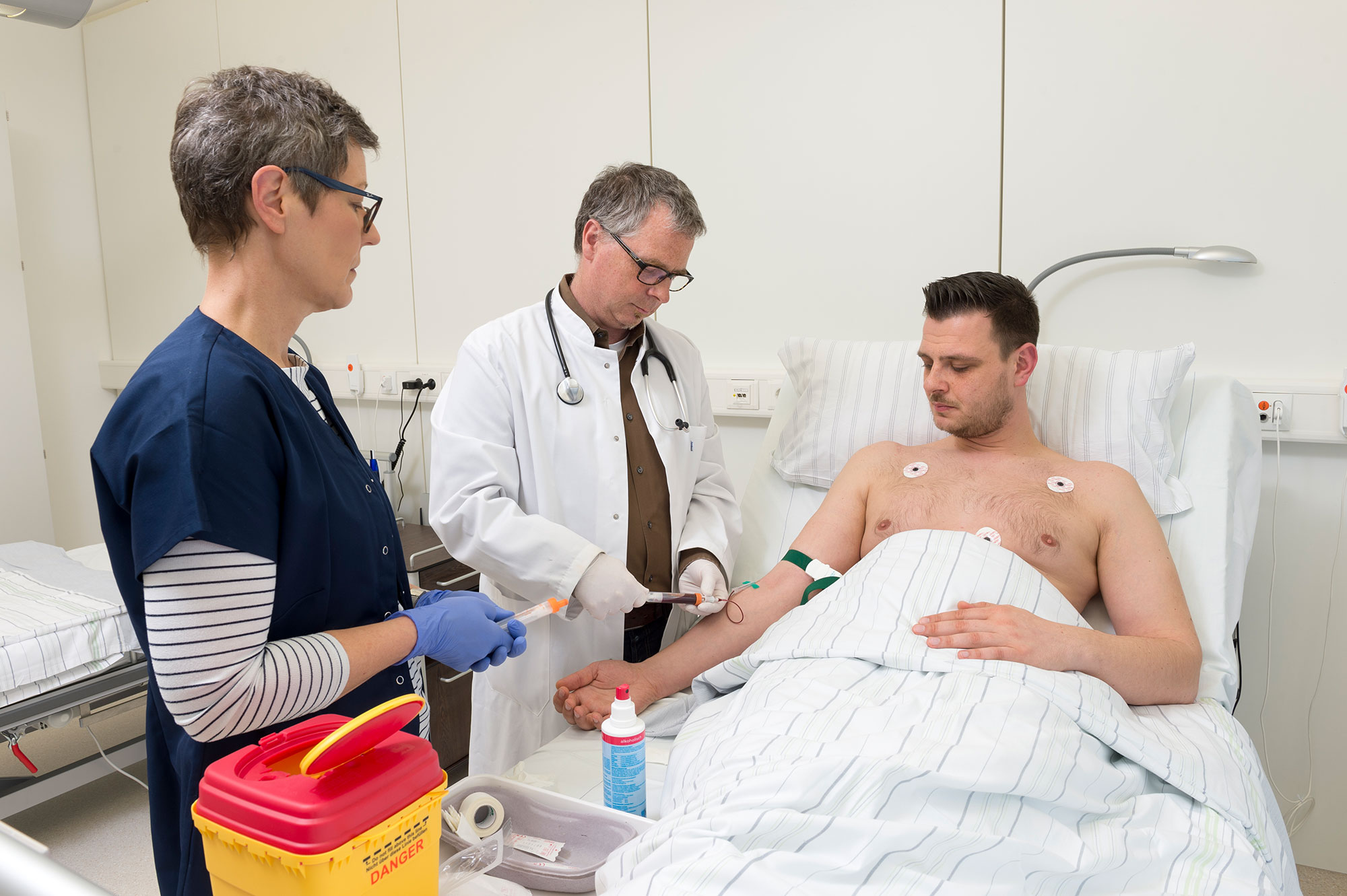
Sabine Schuster* is slightly nervous as she steps into the clinical studies ward run by a Fraunhofer project group called Translational Medicine and Pharmacology TPM on the campus of Frankfurt am Main’s University Hospital. She has never taken part in a drug trial before. As Dr. Jochen Graff, head of the ward, shows Sabine to her room, he explains the drug regulatory agencies’ strict rules in place to ensure trials are standardized.
“You’ll be spending the next two days in our ward with meals and beverages served at set times. You’re not allowed to eat anything else. No cigarettes or alcohol either. That would falsify the results,” says Jochen Graff.
This trial goes to test a new drug for hereditary angioedema (HAE), a rare disease that has plagued Sabine Schuster since childhood. Her skin and internal organs are beset by recurrent swelling. A swollen larynx can be life-threatening; in fact, it is often fatal. Medication is available to manage acute seizures, but this new prophylactic drug aims to prevent these attacks from occurring in the first place. The Fraunhofer project group works closely with the Frankfurt University Hospital’s HAE outpatient unit to conduct clinical trials.
Strict protocol for taking blood samples
The test gets underway the next morning. Research assistant Gudrun Schneckenburger inserts a needle into the vein of Sabine Schuster’s left hand and connects it to the ECG and blood pressure monitor. Clinical investigator Dr. Graff then administers a pill; this tablet contains the new drug.
The research assistant takes blood samples every 15 minutes in the first hour after the tablet is ingested; then every half hour and later every hour. “In the blood samples, we measure what the body does with the drug; how it is absorbed, distributed, broken down and excreted. That is pharmacokinetics,” says Jochen Graff by way of explanation. These readings give scientists insight into the new drug. They also serve to determine doses and dosing intervals for subsequent clinical trials, where the drug has to substantiate its ability to reduce the frequency of seizures in patients with HAE. The researchers assess many different parameters and biomarkers in the blood to identify side effects. Every step of the study is spelled out in a trial protocol that is subject to the approval of the regulatory authorities and an ethics committee.
Innovative study designs
All phases of early clinical drug development take place on the Fraunhofer project group’s ward. This includes phase 1, where the new drug is first administered to humans, and phase 2 with select patients. All phase 1 studies apart from anticancer drug trials have to be conducted with healthy subjects. There is more to these early phases than merely investigating pharmacokinetics and calculating dosages; they also serve to test the active ingredient’s safety.
Dr. Frank Behrens, who heads up the project group’s clinical research, points out the limitations of this approach: “What can a 22-year-old healthy student’s tolerance and metabolism tell me about a drug that I later want to administer to a 65-year-old woman who has already had several prior diseases?” This is why Frank Behrens and his team of scientists from diverse disciplines are developing innovative study designs to test new active ingredients straightaway on patients rather than on healthy subjects. He works closely with medical specialists on the Frankfurt University Hospital campus to select drugs and conduct special tests to prove their effectiveness.
The Frankfurt project group, which is part of the Fraunhofer Institute for Molecular Biology and Applied Ecology IME, has been busy helping set up the LOEWE Research Center for Translational Medicine and Pharmacology funded by the state of Hessen. Its partners are the University of Frankfurt and the Max Planck Institute for Heart and Lung Research in Bad Nauheim. This organization’s mission is to expedite the transition of medical research findings from the test bench to the bedside. Physicians who divide their time between the clinic and the project group play an important role to this end. “Researching physicians bring problems from the patient’s bedside straight to our lab,” emphasizes Prof. Gerd Geisslinger, head of the Fraunhofer project group and the LOEWE Center.
From the lab to the clinic
The aidCURE project shows how R&D plays out all along the value chain in Frankfurt. Determined to develop an active ingredient to treat rheumatoid arthritis in a joint effort with Prof. Rikard Holmdahl of Stockholm’s Karolinska Institute, Prof. Harald Burkhardt spent years conducting fundamental research at the university. This agent is a type of vaccine that targets and switches off the mechanism that triggers pathogenic autoimmune reactions.
The Fraunhofer project group is translating this basic research into applied science under the auspices of Dr. Nadine Schneider. “We are now looking for a way to economically produce the complex protein molecule on an industrial scale,” reports Burkhardt. Clinical testing in humans can commence once the team reaches this milestone and wraps up the preclinical safety studies required by law. Dr. Frank Behrens, head of the study, is already drafting the trial protocols.
A therapy’s success hinges on the diagnosis – the more accurate the disease’s characterization, the more targeted the treatment. Another research team is investigating new diagnostic parameters in a project called RIBULUTION.
*Name changed by the editors
Further information
New Fraunhofer Institute for Translational Medicine and Pharmacology ITMP
In 2012, a Fraunhofer project group for Translational Medicine and Pharmacology TMP was established at the Fraunhofer Institute for Molecular Biology and Applied Ecology IME in association with the LOEWE program (Hessian Initiative for the Development of Scientific-Economic Excellence). The aim of this group was to expand the Fraunhofer-Gesellschaft’s portfolio in the field of drug research and development. Over the last number of years, the former Fraunhofer project group TMP has achieved international recognition in the area of immune disorders due to its extensive expertise in areas such as drug identification, pharmaceutical technology, highly differentiated and indication-specific pharmacological models, and clinical research. As a result of these advances, the TMP branch of Fraunhofer IME is set to become an independent institute with headquarters in Frankfurt am Main and locations in Hamburg and Göttingen as of January 1, 2021.